Ion Channels | Vibepedia
Ion channels are sophisticated pore-forming proteins embedded within cell membranes, acting as highly selective conduits for the passage of charged ions like…
Contents
- 🎵 Origins & History
- ⚙️ How It Works
- 📊 Key Facts & Numbers
- 👥 Key People & Organizations
- 🌍 Cultural Impact & Influence
- ⚡ Current State & Latest Developments
- 🤔 Controversies & Debates
- 🔮 Future Outlook & Predictions
- 💡 Practical Applications
- 📚 Related Topics & Deeper Reading
- Frequently Asked Questions
- References
- Related Topics
Overview
The concept of ion channels emerged from early 20th-century studies on nerve impulse transmission. Pioneers like Alan Hodgkin and Andrew Huxley, through their groundbreaking work on the squid giant axon in the 1930s and 1940s, proposed a model involving transient changes in membrane permeability to sodium and potassium ions, laying the theoretical groundwork for voltage-gated channels. The direct visualization and functional characterization of individual ion channels became possible with the development of the patch-clamp technique by Erwin Neher and Bert Sakmann in the late 1970s, for which they received the Nobel Prize in Physiology or Medicine in 1991. This technique allowed researchers to isolate and record the tiny electrical currents flowing through single channels, transforming the field from theoretical models to direct molecular observation. Subsequent decades saw the cloning and sequencing of genes encoding numerous ion channel families, revealing their diverse structures and evolutionary relationships.
⚙️ How It Works
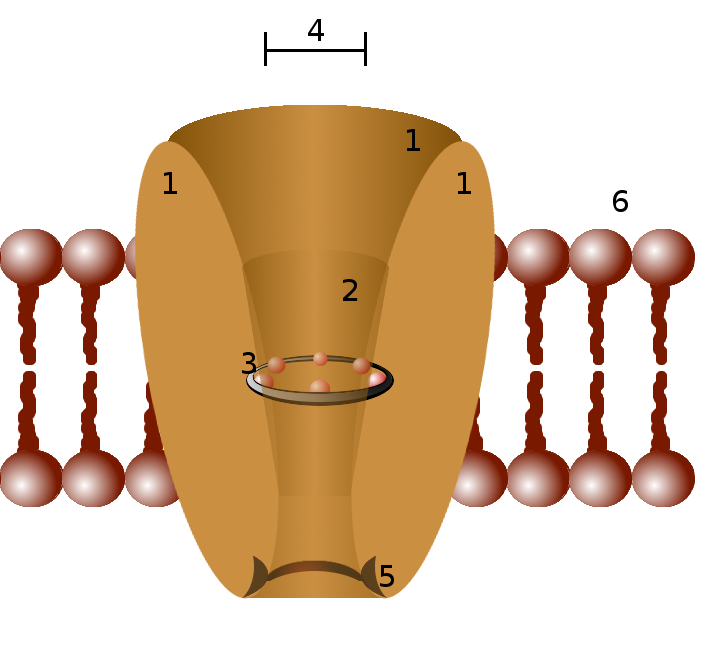
Ion channels function as selective pores through the lipid bilayer of cell membranes, facilitating the rapid, passive movement of specific ions down their electrochemical gradients. Each channel protein typically consists of multiple subunits that assemble to form a central pore. The 'selectivity filter,' a narrow region within the pore, is lined with amino acid residues that precisely interact with specific ions, allowing passage of certain ions while excluding others based on size, charge, and hydration. The 'gate' is a dynamic part of the channel that can open or close in response to various stimuli, such as changes in membrane potential (voltage-gated channels), binding of specific ligands like neurotransmitters (ligand-gated channels), mechanical stress (mechanically-gated channels), or intracellular signaling molecules (e.g., cyclic AMP-gated channels). This regulated opening and closing controls ion flux, thereby shaping cellular electrical activity and mediating signal transduction.
📊 Key Facts & Numbers
There are estimated to be over 300 different types of ion channels in the human genome, belonging to at least 10 major superfamilies. These channels are responsible for generating the resting membrane potential in virtually all cells, which typically ranges from -40 mV to -80 mV. For instance, the resting membrane potential of neurons is largely determined by the selective permeability to potassium ions through leak channels. Action potentials, the electrical signals used by neurons and muscle cells, involve rapid, transient changes in membrane permeability to sodium and potassium ions, with durations often measured in milliseconds. Calcium channels play a critical role in neurotransmitter release, with influxes as brief as 100 microseconds triggering vesicle fusion. The collective activity of ion channels underlies an estimated 10-20% of all human genes encoding membrane proteins.
👥 Key People & Organizations
Key figures in ion channel research include Alan Hodgkin and Andrew Huxley, whose mathematical model of nerve impulse propagation in 1952 remains a foundational text. Erwin Neher and Bert Sakmann revolutionized the field with their development of the patch-clamp technique in the late 1970s, enabling the study of single ion channels. Roderick MacKinnon was awarded the Nobel Prize in Chemistry in 2003 for his work on the structure and mechanism of potassium channels, including the first atomic resolution structure of a potassium channel. Major research institutions like The Rockefeller University, Stanford University, and the Max Planck Society host leading ion channel research labs. Pharmaceutical companies such as Pfizer, Merck, and Novartis invest heavily in ion channel drug discovery.
🌍 Cultural Impact & Influence
Ion channels are central to the electrical excitability of cells, forming the basis of nerve signaling, muscle contraction, and cardiac rhythm. Their role in neurotransmission makes them critical for cognition, learning, and behavior, influencing everything from memory formation to mood regulation. The precise control of ion flux by channels also underpins sensory perception, such as vision, hearing, and touch. Beyond excitable cells, ion channels regulate epithelial transport in organs like the kidneys and lungs, impacting fluid balance and respiration. Their widespread influence has made them a recurring motif in scientific literature and popular science, often depicted as the 'doors' or 'gates' that control cellular communication and function. The discovery of specific channelopathies, diseases caused by ion channel dysfunction, has further cemented their importance in understanding human health and disease.
⚡ Current State & Latest Developments
Current research is rapidly advancing our understanding of ion channel function, particularly through cryo-electron microscopy (cryo-EM) and advanced computational modeling, which are revealing channel structures in unprecedented detail. The development of novel pharmacological tools, including highly selective small molecules and gene therapies, is opening new avenues for treating diseases linked to ion channel dysfunction. For example, recent breakthroughs are exploring gene editing techniques like CRISPR-Cas9 to correct mutations in ion channel genes responsible for conditions like cystic fibrosis (CFTR channels) and certain forms of epilepsy. The field of 'channelomics' is also expanding, aiming to catalog and functionally characterize the entire complement of ion channels in various cell types and tissues, providing a systems-level view of their roles in health and disease.
🤔 Controversies & Debates
A significant debate revolves around the precise mechanisms of channel gating and ion permeation, particularly for complex channels with multiple gating mechanisms or unusual ion selectivity. The 'selectivity filter' remains an area of intense study, with ongoing research to fully elucidate how channels achieve such exquisite specificity. Another controversy concerns the development of drugs targeting ion channels; while many diseases are linked to channel dysfunction, achieving therapeutic selectivity without causing off-target effects (e.g., cardiac arrhythmias from potassium channel blockers) remains a major challenge. Furthermore, the role of 'silent' or poorly characterized ion channels in disease pathogenesis is increasingly recognized, sparking debate about their potential as therapeutic targets.
🔮 Future Outlook & Predictions
The future of ion channel research is poised for significant advancements. We can expect the development of even more precise tools for manipulating and observing ion channel activity in living systems, potentially including nanoscale biosensors and advanced optogenetic techniques. Personalized medicine will increasingly leverage our understanding of individual genetic variations in ion channels to tailor drug treatments for channelopathies. The integration of artificial intelligence and machine learning with large-scale ion channel data (channelomics) is likely to accelerate drug discovery and the identification of novel therapeutic targets. Furthermore, research into the role of ion channels in non-canonical functions, such as cell proliferation and cancer, is expected to expand, potentially revealing new therapeutic strategies for oncological diseases.
💡 Practical Applications
Ion channels are indispensable in numerous practical applications. In medicine, they are targets for a vast array of drugs used to treat conditions such as hypertension (calcium channel blockers like amlodipine), epilepsy (sodium channel blockers like phenytoin), pain (TRP channel modulators), and cardiac arrhythmias (potassium channel blockers). In biotechnology, ion channels are used in biosensors for detecting specific molecules or environmental conditions. In research, they are crucial tools for understanding cellular physiology and for developing new therapeutic strategies. For example, the CFTR channel modulator ivacaftor has dramatically improved outcomes for patients with certain forms of cystic fibrosis. The development of artificial ion channels is also an emerging area with potential applications in desalination and energy production.
Key Facts
- Year
- 1952 (theoretical basis)
- Origin
- Global
- Category
- science
- Type
- concept
Frequently Asked Questions
What exactly is an ion channel and how does it work?
An ion channel is a protein embedded in a cell's membrane that forms a pore, allowing specific charged ions (like sodium, potassium, calcium, or chloride) to pass through. It works by selectively binding and transporting these ions down their concentration and electrical gradients. The channel's 'gate' can open or close in response to various signals, such as voltage changes across the membrane, binding of signaling molecules, or physical pressure, thereby controlling the flow of ions and influencing cellular activity.
Why are ion channels so important for the human body?
Ion channels are critical for virtually all physiological functions. They generate the electrical signals that allow neurons to communicate (action potentials), muscles to contract, and the heart to beat. They also regulate the movement of ions and water across epithelial tissues, control hormone secretion, and are involved in sensory perception like taste and pain. Without functional ion channels, basic cellular processes and complex bodily functions would cease.
What happens when ion channels don't work properly?
When ion channels malfunction due to genetic mutations or other factors, it leads to a class of diseases known as channelopathies. These can manifest in a wide range of symptoms depending on the affected channel and tissue. Examples include epilepsy (faulty sodium or calcium channels in neurons), cystic fibrosis (defective CFTR chloride channel), certain types of inherited deafness or blindness, cardiac arrhythmias, and chronic pain conditions. The impact can be severe, affecting neurological, muscular, cardiovascular, and other organ systems.
How do scientists study ion channels?
Scientists use a variety of sophisticated techniques to study ion channels. The gold standard is the patch-clamp technique, which allows researchers to record the electrical current flowing through single ion channels or whole cell populations. Other methods include voltage-gated fluorescence imaging, genetic analysis to identify channel genes and mutations, X-ray crystallography and cryo-electron microscopy to determine their 3D structures, and pharmacological studies using specific drugs to probe channel function.
Are ion channels a target for new medicines?
Absolutely. Ion channels are among the most successful targets for drug development, with numerous approved medications acting on them. For instance, calcium channel blockers are widely used for hypertension, sodium channel blockers for epilepsy and arrhythmias, and potassium channel modulators for conditions like cystic fibrosis. The ongoing discovery of new channel types and their roles in disease continues to fuel pharmaceutical research, making them a vital area for developing treatments for neurological disorders, cardiovascular diseases, pain, and more.
Can we create artificial ion channels?
Yes, researchers are actively working on creating artificial ion channels. These synthetic channels, often inspired by natural ion channel structures or using novel materials like engineered peptides or polymers, aim to mimic the selectivity and gating properties of biological channels. Potential applications include advanced water purification (desalination), energy generation, and novel drug delivery systems. While still largely in the research phase, artificial ion channels represent a promising frontier in nanotechnology and biomaterials science.
What is 'channelomics'?
'Channelomics' is a field that aims to comprehensively catalog, characterize, and understand the function of all ion channels within a cell, tissue, or organism. It involves integrating data from genomics, transcriptomics, proteomics, and electrophysiology to build a systems-level view of ion channel activity. The goal is to map the complete 'ion channel landscape' to better understand their roles in health and disease and to identify new therapeutic targets by analyzing how the entire set of channels contributes to cellular and organismal function.