Antibiotic Therapy | Vibepedia
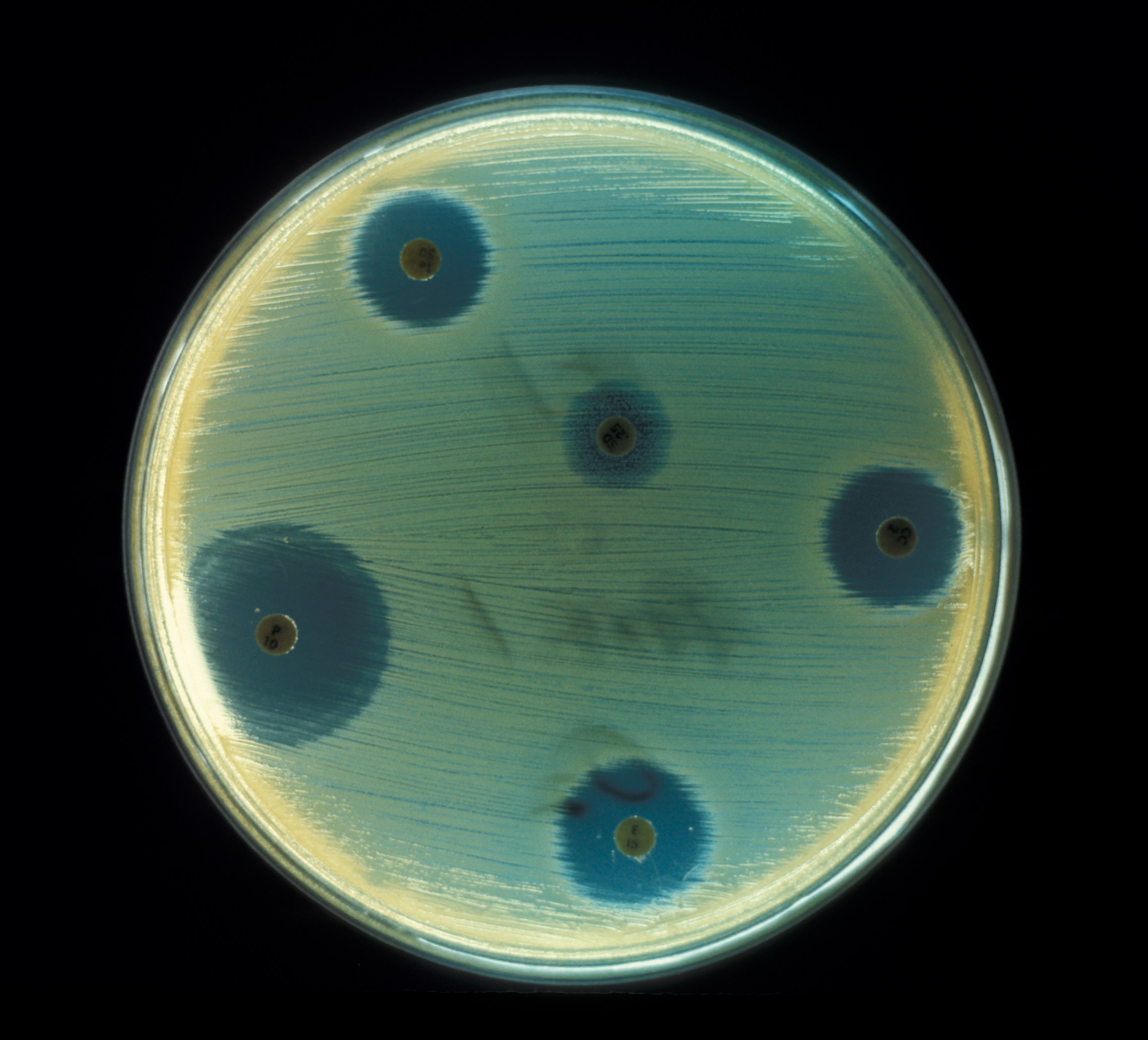
Antibiotic therapy is the cornerstone of modern medicine, employing drugs to combat bacterial infections by either killing bacteria (bactericidal) or…
Contents
- 🎵 Origins & History
- ⚙️ How It Works
- 📊 Key Facts & Numbers
- 👥 Key People & Organizations
- 🌍 Cultural Impact & Influence
- ⚡ Current State & Latest Developments
- 🤔 Controversies & Debates
- 🔮 Future Outlook & Predictions
- 💡 Practical Applications
- 📚 Related Topics & Deeper Reading
- Frequently Asked Questions
- References
- Related Topics
Overview
The genesis of antibiotic therapy is inextricably linked to the accidental observation of penicillin's antibacterial properties by Alexander Fleming in his St. Mary's Hospital laboratory in London in 1928. Fleming noted that a mold, later identified as Penicillium notatum, inhibited the growth of Staphylococcus bacteria. While Fleming's discovery was groundbreaking, it was the subsequent work of Howard Florey and Ernst Chain at the University of Oxford in the early 1940s that led to the purification and mass production of penicillin, making it a viable therapeutic agent during World War II. This era marked the dawn of the 'antibiotic age,' ushering in a cascade of discoveries, including streptomycin by Selman Waksman in 1943, which proved effective against tuberculosis. The initial success and widespread adoption of these early antibiotics created a profound sense of optimism, leading many to believe infectious diseases would soon be eradicated.
⚙️ How It Works
Antibiotic therapy functions by targeting specific biochemical pathways essential for bacterial survival and replication, pathways that are either absent or significantly different in human cells, thus minimizing host toxicity. Broadly, antibiotics are classified by their mechanism of action: cell wall synthesis inhibitors (like penicillin and cephalosporins), protein synthesis inhibitors (such as tetracyclines and macrolides), nucleic acid synthesis inhibitors (e.g., quinolones and rifampicin), and metabolic pathway inhibitors (like sulfonamides). The choice of antibiotic depends on the identified or suspected pathogen, its susceptibility profile, the site of infection, and patient-specific factors like allergies and renal function. This targeted approach allows for the selective elimination of harmful bacteria while preserving the host's own microbial flora, though disruption of this flora can lead to secondary issues like Clostridioides difficile infections.
📊 Key Facts & Numbers
The global antibiotic market was valued at approximately $45 billion in 2022, with projections indicating steady growth, albeit tempered by resistance concerns. Over 100 different antibiotic drugs are currently approved for human use, categorized into various classes and generations. In the United States alone, it's estimated that over 270 million antibiotic prescriptions were dispensed in outpatient settings in 2020, with a significant portion deemed unnecessary. Globally, an estimated 1.27 million deaths in 2019 were directly attributable to bacterial infections resistant to antibiotics, a figure projected to rise dramatically to 10 million deaths annually by 2050 if current trends persist. The development cost for a new antibiotic can exceed $1 billion, a significant barrier to innovation, with many pharmaceutical companies exiting the field due to poor return on investment compared to chronic disease medications.
👥 Key People & Organizations
Key figures in the history of antibiotic therapy include Alexander Fleming, whose discovery of penicillin in 1928 laid the foundation for the field. Howard Florey and Ernst Chain were instrumental in developing penicillin into a usable drug, earning them a Nobel Prize alongside Fleming. Selman Waksman, a Ukrainian-American biochemist, discovered streptomycin in 1943, the first effective treatment for tuberculosis, and coined the term 'antibiotic.' Major organizations driving research and policy include the World Health Organization (WHO), which spearheads global strategies against antimicrobial resistance, and the U.S. Food and Drug Administration (FDA), which approves new antibiotic drugs. Pharmaceutical giants like Pfizer, Merck, and Novartis have historically been major players in antibiotic development, though many are now shifting focus due to economic pressures.
🌍 Cultural Impact & Influence
Antibiotic therapy has profoundly reshaped human civilization, enabling complex surgeries, organ transplantation, and chemotherapy by mitigating the risk of infection. Before antibiotics, common bacterial infections like pneumonia or sepsis were often death sentences, and childbirth carried a high risk of fatal puerperal fever. The widespread availability of antibiotics in the mid-20th century contributed significantly to increased life expectancy and population growth. Culturally, antibiotics became symbols of medical progress and scientific triumph, often depicted in media as miracle cures. However, this success also fostered a sense of complacency, leading to the perception of antibiotics as a panacea for all infections, including viral ones, contributing to their overuse and the subsequent rise of resistance, a narrative explored in works like the documentary 'Resistance'.
⚡ Current State & Latest Developments
The current state of antibiotic therapy is characterized by a critical tension between ongoing innovation and the relentless march of antimicrobial resistance. While new antibiotic classes are scarce, research is exploring novel approaches, including phage therapy, antimicrobial peptides, and microbiome-based interventions. In 2023, the FDA approved several new antibiotics, such as lefamulin for community-acquired bacterial pneumonia and plazomicin for complicated urinary tract infections, offering hope against specific resistant pathogens. Simultaneously, global surveillance efforts, like those coordinated by the World Health Organization, are intensifying to track resistance patterns and inform public health responses. The push for antibiotic stewardship programs in hospitals and communities worldwide aims to optimize antibiotic use, reserving potent agents for when they are truly necessary and ensuring patients complete their prescribed courses.
🤔 Controversies & Debates
The most significant controversy surrounding antibiotic therapy is the escalating crisis of antimicrobial resistance. Critics argue that pharmaceutical companies have underinvested in new antibiotic development due to poor profitability, leading to a 'market failure' that jeopardizes global health security. Public health advocates and organizations like the World Health Organization decry the widespread overuse and misuse of antibiotics in both human medicine and agriculture, where they are often employed for growth promotion rather than treating infections. Ethical debates also arise concerning access to essential antibiotics in low-resource settings and the potential for antibiotic resistance to exacerbate existing health inequities. The development of new antibiotics is often hampered by regulatory hurdles and the lengthy, expensive clinical trial process.
🔮 Future Outlook & Predictions
The future of antibiotic therapy hinges on a multi-pronged strategy to combat resistance and revitalize drug development. Experts predict a rise in 'last-resort' antibiotics, requiring stringent usage protocols and potentially higher costs. Innovations like CRISPR-based gene editing are being explored to directly target resistance genes within bacteria. The development of vaccines against common bacterial pathogens, such as pneumococcal and meningococcal vaccines, offers a proactive approach to reducing the need for antibiotic treatment. Furthermore, a greater emphasis on diagnostics to rapidly identify pathogens and their resistance profiles will enable more precise and effective therapy. International collaborations and novel economic models, such as subscription-based payment systems for antibiotics, are being proposed to incentivize pharmaceutical investment in this critical area.
💡 Practical Applications
Antibiotic therapy is indispensable across numerous medical disciplines. In surgery, prophylactic antibiotics are routinely administered to prevent post-operative infections, particularly in procedures like joint replacements or abdominal surgeries. Oncology relies heavily on antibiotics to manage infections in immunocompromised patients undergoing chemotherapy. Intensive care units frequently use broad-spectrum antibiotics to treat severe sepsis and hospital-acquired infections. In veterinary medicine, antibiotics are crucial for treating infections in livestock and companion animals, though their use in food-producing animals is increasingly scrutinized due to resistance concerns. Public health initiatives focus on promoting appropriate antibiotic use through education campaigns and prescription guidelines.
Key Facts
- Year
- 1928-present
- Origin
- United Kingdom
- Category
- science
- Type
- concept
Frequently Asked Questions
What is the difference between an antibiotic and an antiviral?
Antibiotics are specifically designed to kill or inhibit the growth of bacteria, targeting unique bacterial cellular processes. Antivirals, on the other hand, are used to treat viral infections by interfering with the virus's ability to replicate within host cells. Antibiotics have no effect on viruses, and antivirals are ineffective against bacteria. For instance, penicillin is an antibiotic used for strep throat, while oseltamivir (Tamiflu) is an antiviral used for influenza.
How does antibiotic resistance develop?
Antibiotic resistance arises through natural selection. When bacteria are exposed to an antibiotic, susceptible bacteria are killed, but any bacteria possessing genetic mutations that confer resistance can survive and multiply. Over time, these resistant strains become dominant. This process is accelerated by the overuse and misuse of antibiotics in human medicine, agriculture, and the environment, providing more opportunities for resistance to emerge and spread globally through mechanisms like horizontal gene transfer between bacteria.
Why are new antibiotics not being developed as quickly as before?
Developing new antibiotics is a complex, expensive, and high-risk endeavor with a low return on investment compared to drugs for chronic conditions. The short treatment duration for antibiotics and the need to reserve new drugs to combat resistance mean they are used sparingly, limiting sales. Furthermore, the increasing prevalence of resistance means that even newly developed drugs may have a limited lifespan. This economic reality has led many major pharmaceutical companies to exit the antibiotic development market, creating a critical gap in the pipeline.
What is antibiotic stewardship?
Antibiotic stewardship refers to coordinated interventions designed to optimize the use of antibiotics and combat resistance. It involves promoting the selection of the most appropriate antibiotic, dose, route, and duration of therapy. Key components include educating healthcare providers and patients, implementing guidelines for antibiotic prescribing, using diagnostic tests to identify pathogens and resistance patterns, and monitoring antibiotic use. Hospitals and health systems worldwide are implementing stewardship programs to ensure antibiotics are used effectively and responsibly.
Can antibiotics be used to treat non-bacterial infections?
No, antibiotics are ineffective against non-bacterial infections such as those caused by viruses, fungi, or parasites. Prescribing antibiotics for conditions like the common cold, flu, or most sore throats (which are typically viral) is not only useless but also harmful. This inappropriate use contributes to the development of antibiotic resistance, making it harder to treat actual bacterial infections when they occur. It also exposes patients to unnecessary side effects and increases healthcare costs.
What are the main classes of antibiotics and how do they work?
Major antibiotic classes include beta-lactams (like penicillin and cephalosporins), which inhibit bacterial cell wall synthesis; macrolides (e.g., azithromycin), which inhibit protein synthesis; fluoroquinolones (e.g., ciprofloxacin), which interfere with DNA replication; and sulfonamides (e.g., trimethoprim-sulfamethoxazole), which block folic acid synthesis. Each class targets a different essential bacterial process, leading to either bacterial death (bactericidal) or growth inhibition (bacteriostatic).
What is the future of antibiotic therapy in the face of resistance?
The future involves a multi-pronged approach: developing novel antibiotics with new mechanisms of action, exploring alternative therapies like phage therapy and antimicrobial peptides, enhancing vaccines to prevent bacterial infections, and implementing robust global antibiotic stewardship programs. Precision medicine, guided by rapid diagnostics, will play a crucial role in tailoring treatments. International collaboration and new economic incentives are vital to encourage pharmaceutical investment in this critical area to avert a post-antibiotic era.